Driving Patient-Centered Innovation Within Regulatory Frameworks
Key Takeaways
- Rapid Growth of the Market: Virtual clinical trials are expected to achieve $11.49B by 2032 as a result of access, convenience, and technology.
- Flexibility and Patient Convenience: Virtual trial models enhance recruitment and retention of patients by removing geographic, socio-economic, and logistical barriers.
- Compliance Hurdles: There are several challenges that remain, including data privacy (GDPR, HIPAA), their fragmented and diverse regulatory environments, and data quality in remote environments.
- Hybrid and AI Enhanced Solutions: Hybrid trial designs and AI-enabled technologies can help balance convenience with compliance by improving recruitment and data monitoring, while optimizing operations through enhancements to patient experience and operational feasibility.
- Future Success Aspect: The success of virtual clinical research in the long term is purely dependent on being inclusive and interoperable. Addressing the digital divide, improving access and integrating technology into the healthcare landscape, and achieving global harmonization of regulatory structures will be the focus of the future of clinical trials.
The New Clinical Trial Landscape
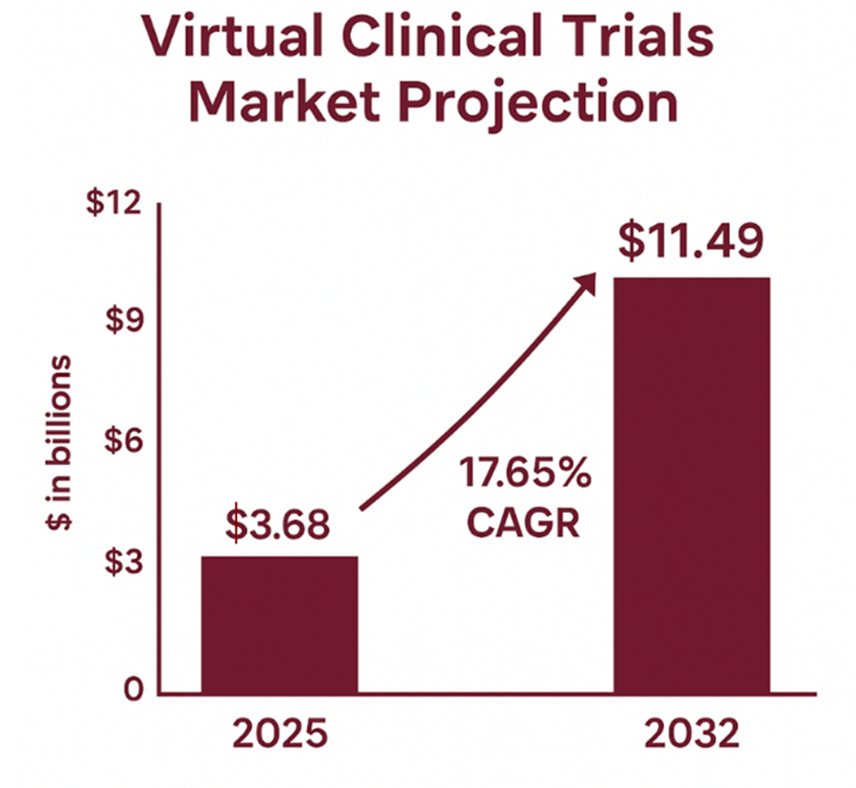
The clinical research industry is changing at an unprecedented rate, particularly due to the COVID-19 pandemic and technology fast tracking innovation. Virtual clinical trials, sometimes referred to as decentralized or hybrid clinical trials are one of the largest changes in medical research methodology since the inception of the randomized controlled trial. The market valuation for virtual clinical trials is projected to be $3.68 billion in 2025, and to reach $11.49 billion by 2032 with a compound annual growth rate of 17.65%. This rapid growth reflects the acceptance of new and innovative ways to conduct trials and the efficiencies they provide. Although sponsors are becoming accustomed to the incredible convenience of virtual trials, regulatory compliance and data protection are responsibilities sponsors must continue to take seriously and still have to navigate thoughtfully.
The Convenience Revolution: Expanding Access and Participation
Virtual clinical trials offer convenience to participants that can radically change strategies for patient recruitment and retention. These trials may open wider and more diverse patient populations, as the participation demands are reduced and the geographical barriers removed. Clinical trials want to identify participants who match the broader demographic of the patient population they represent. That means, traditionally, to participate in a clinical trial, participants need to live in the proximity of research sites, which presents limitations on demographics. Virtual trials can reach participants over larger geographical distances, including areas where rural and underserved populations have not had research access.
The convenience factor extends far beyond geography. Virtual trials offer a participant the ability to be involved in research during their own time; they don’t have to take time off work, get transport arranged, or source childcare. McKinsey research on this topic highlights how the Covid-19 pandemic catalyzed unprecedented uptake of decentralized models, as trial sites were unreachable and sponsors had to adopt remote consent, video-consent and remote phlebotomy. Post- pandemic findings show that 98.7% of patients reported high levels of satisfaction with their telemedicine components of care, while 72.8% of physicians reported a similar/better experience with remote engagement compared to in-person.
The Compliance Imperative: Navigating Regulatory Complexities
While the potential advantages of convenience are undeniable, virtual trials come with challenging compliance hurdles that sponsors will have to manage. Regulations such as the General Data Protection Regulation (GDPR) in Europe and the Health Insurance Portability and Accountability Act (HIPAA) in the United States impose strict obligations related to sensitive health information. The regulations require robust data protection, including required explicit informed consent to use the data, secure encryption, and ensuring access control. Many trials are multinational, and this creates additional layers of complexity as sponsors must navigate the multitude of regulations.
Agencies that serve to regulate use, and thus must have a consideration for technology innovation. The FDA has expanded its digital trial guidance, while the EMA is advocating for a common framework for decentralized trials in Europe. Despite discussions of synergies and integration, the regulatory landscape is still disjointed, with various agencies and countries embracing different levels of virtual trial elements. It creates compliance challenges for sponsors when they operate in more than one Jurisdiction.
| Consideration | Challenge | Emerging Solutions |
| Data Privacy | Ensuring compliance with GDPR, HIPAA, and other regional regulations | Privacy-by-design IT systems, anonymization techniques, and secure data transmission protocols |
| Regulatory Harmonization | Differing requirements across countries and regions | Regulatory sandboxes, early engagement with health authorities, and adaptive trial designs |
| Technical Validation | Demonstrating that digital endpoints meet regulatory standards for validity | Pre-trial validation studies, use of FDA-approved devices, and robust technical documentation |
| Patient Comprehension | Ensuring proper understanding of consent processes in remote settings | Multimedia consent materials, interactive consent platforms, and comprehension assessments |
| Data Quality | Maintaining data accuracy without direct site supervision | Remote monitoring technologies, automated data checks, and patient training programs |
Striking the Balance: Integrating Convenience with Compliance
Whether the future of virtual trials can benefit from transformed conveniences and provide robust mechanisms for compliance will depend on taking a considered approach to trial design, technology, and operational delivery. Hybrid trial designs have emerged, combining aspects of traditional trials with decentralized trials, and are an exciting option. Hybrid trials preserve site-based activities when sponsors or study teams need to retain some complex or invasive procedures performed at the site, where certain study processes can be decentralized and completed most effectively at a remote location. Careful planning and design of the trial may then optimize the aspects of convenience while limiting compliance risk.
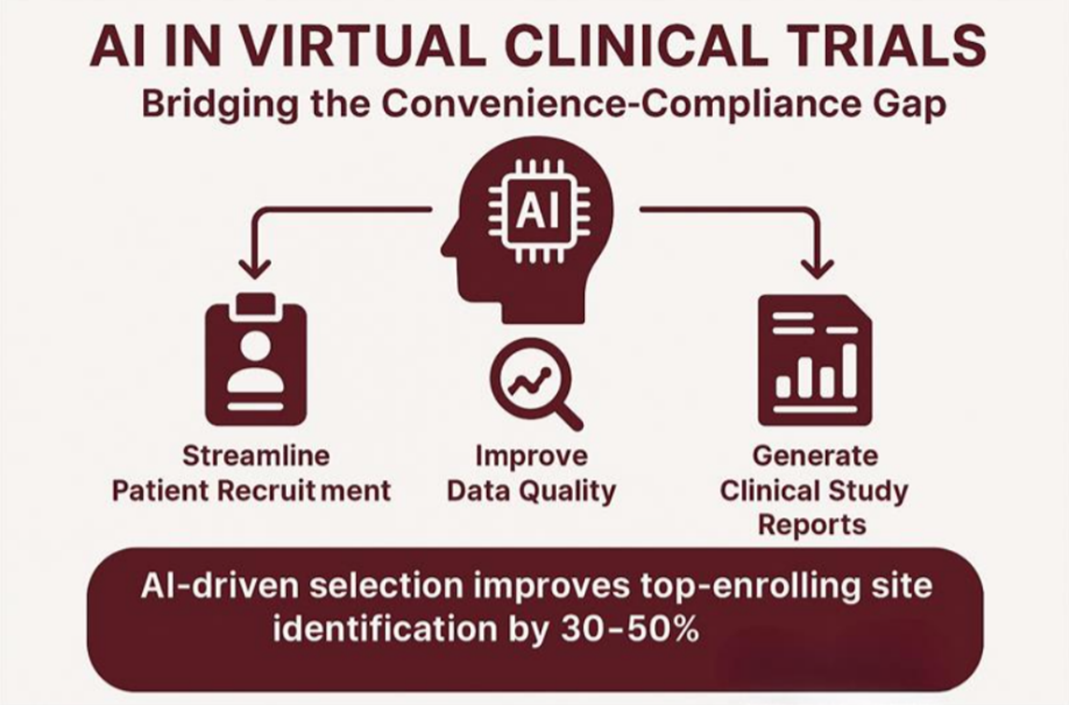
Technology is a critical factor in filling the convenience-compliance gap. Artificial Intelligence (AI) and machine learning technologies are being more frequently used to enhance both convenience and compliance for virtual trials. AI is used to predict and automate patient recruitment, improve data quality with automated monitoring, and improve clinical study reports with 98% accuracy. McKinsey notes, “AI-enabled selection increases the identification of best-enrolling sites by 30-50% and shortens enrollment time by 10 to 15%“. These technologies enhance not only convenience but compliance with enhanced data management and documentation.
The Path Forward: Key Considerations for Future Success
As virtual trials continue to develop, sponsors will need to balance convenience with compliance by addressing three key challenges.
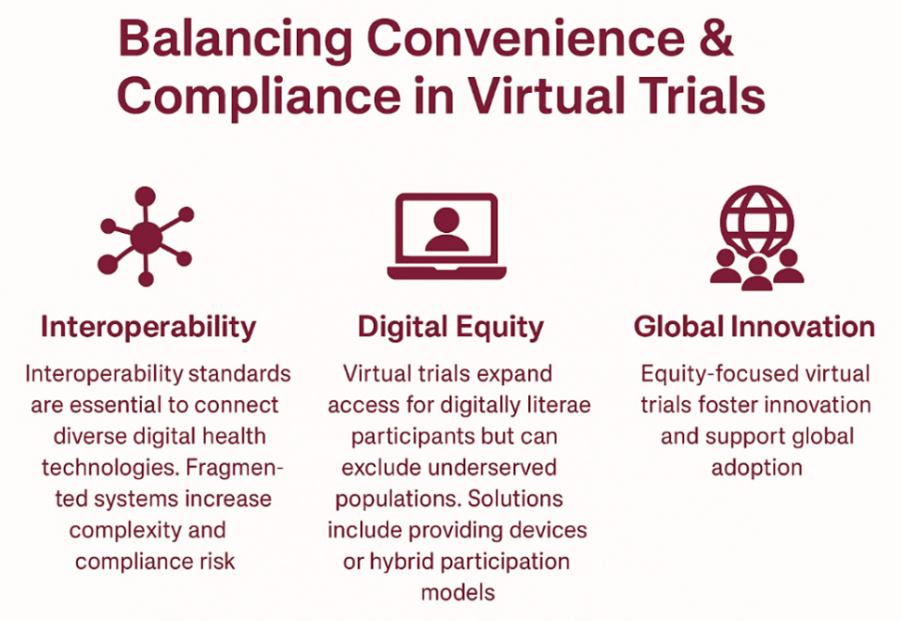
First, we need interoperability standards to connect a wide variety of digital health technologies with trial infrastructures. The more fragmented the systems across trial sites and studies, the more complexity and compliance risk are introduced into the trial process.
Second, although virtual trials can expand access to trials for those who are digitally literate, they can also limit access for people in underserved areas without reliable internet connections, devices, or comfort with digital technology and interfaces. Sponsors can ramp up equity by providing access to devices or flexible hybrid models that allow participants to complete activities in the way that is most convenient for them. Sponsoring virtual trials in ways that put equity at the forefront will allow for innovation and support global uptake.
Finally, regulatory consistency is extremely important and provides scalability across jurisdictions. The FDA and EMA both released guidance for decentralized trials, but significant variability still exists. To ensure regulatory compliance, sponsors will need to interact with regulators throughout their trial networks early and often. Addressing these challenges provides a pathway to not just more effective trials, but more equitable, virtual trials on a global level.
Conclusion: Achieving Synergy Between Convenience and Compliance
The future of virtual clinical trials lies not in opting for convenience over compliance, but rather in integrating both. The convenience of decentralized approaches could revolutionize clinical research, providing much more access to diverse populations, as well as minimizing the burdens that have historically inhibited recruitment and retention. However, compliance with regulatory expectations and data protection standards to ensure patient safety and data integrity is equally important for securing regulatory approval. As the market continues to evolve, those sponsors that can best manage the tension between these competing priorities will realize considerable improvements in both trial efficiency and effectiveness. By applying patient-centric design, implementing strong technology consideration, and creating a collaborative relationship with regulators, sponsors will be able to maximize the benefits of virtual trials without compromising compliance or quality. Ultimately, the research ecosystem will become more effective and more inclusive, benefitting sponsors, researchers, and patients.
Recommendation For Stakeholders
- Invest in Regulatory Strategy: Start early by having pre-submission meetings with regulatory agencies (FDA, EMA) to ensure clarity around decentralized or hybrid trial components, and be proactive by regularly following up on updates to regulatory guidance to adapt trial protocols in advance and avoid delays.
- Adopt Hybrid Trial Models: Strategically allocate complex procedures requiring specialized equipment or supervision to on-site visits, while assigning simple tasks (surveys, routine monitoring) remotely, and allow participants to choose remote or on-site participation options where feasible to increase convenience and adherence.
- Prioritize Patient Education & Support: Use interactive tools such as videos, infographics, or e-consent portals to enhance clarity and engagement in trial instructions, while providing ongoing support through helplines, chat support, or “participant rally” sessions to address questions and reassure adherence to protocol.
- Strengthen Data Quality Controls: Undergo rigorous pre-trial testing of digital devices for reliability and regulatory compliance, while leveraging automated alerts and data checks to quickly identify anomalies and protect scientific integrity.
- Address the Digital Divide: Provide necessary hardware, internet connections, or technical support to participants lacking digital resources, while offering a mix of in-person and remote participation options to include diverse population groups.

